 It’s another busy day in Virchester and you are put on standby for a 68 year old male who has had an out of hospital cardiac arrest. Fortunately it was a witnessed arrest and there was good bystander CPR. The patient had early defibrillation from the a first aider trained in the use of an automatic defib. The paramedics arrived soon after to confirm a return of spontaneous circulation (ROSC) after about 10 minutes of downtime with good CPR throughout. The initial rhythm is not clear, but since the AED gave a shock you presume that it was VF or VT.
It’s another busy day in Virchester and you are put on standby for a 68 year old male who has had an out of hospital cardiac arrest. Fortunately it was a witnessed arrest and there was good bystander CPR. The patient had early defibrillation from the a first aider trained in the use of an automatic defib. The paramedics arrived soon after to confirm a return of spontaneous circulation (ROSC) after about 10 minutes of downtime with good CPR throughout. The initial rhythm is not clear, but since the AED gave a shock you presume that it was VF or VT.
You do a quick ABC assessment. The patient is still a little confused, but GCS 14. They don’t have chest pain at the moment. Obs are pretty good with a satisfactory BP and a heart rate of 70 with normal sinus rhythm.
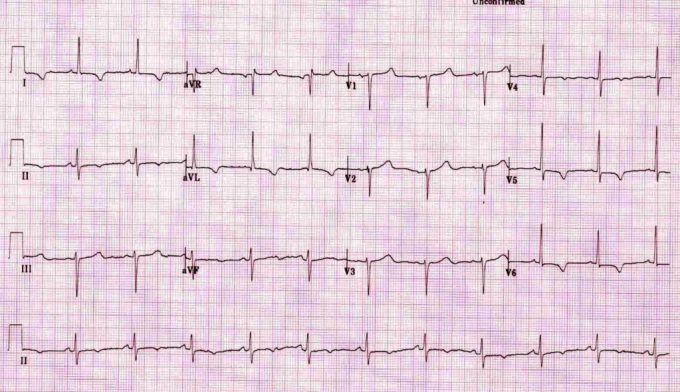
You get lines in, get some bloods off, check a gas (it’s not that bad) and have a look at the 12 lead ECG (taken from the ECG library at LITFL).
You discuss with a friend and you both agree it’s not normal, but there is no ST elevation to confirm an AMI. Should he go to the cath lab? If it’s ST elevation on the ECG then it’s clear and agreed, off to the cath lab they go1,2,3 You’re not sure of the local protocols for non ST elevation patients and so give the on call cardiology team a ring. They decline the patient, suggesting aspirin, LMWH and Ticagrelor and an admission under the acute physicians. They roll their eyes and bounce it back to cardiology in the never ending game of patient ping-pong. The patient finally gets admitted to coronary care, has a huge troponin rise and thus gets a cardiac catheterisation with stents the next day.
You reflect on this and wonder whether these patients should just go to the cath lab regardless of that initial ECG.
You flick through your emails and notice this rather interesting paper that might just answer your question4.
 It looks as though this may answer the question so read on.
It looks as though this may answer the question so read on.
What kind of paper is this?
It’s a systematic review and meta-analysis. In the hierachy of evidence that’s almost at the top (an n of 15 is actually the highest EBM level). However, that’s just a description of paper type. All too often we see clinicians just assume that the conclusions from a meta-analysis are correct without critically appraising the review itself. This is a real error as a review may also be prone to bias and error. We must always review the review!
What was studied?
Arguably the ‘subjects’ of a review are the papers used to come to a conclusion. We need to know how and why the papers were selected and whether they were subject to a structured critical appraisal. In this paper they searched several databases, the grey literature, refererences and conference reports. This is pretty comprehensive and it’s unlikely that they missed any significant papers.
In terms of quality then they applied a methodological filter, excluding lower levels of evidence such as case reports and letters, and then also used an appraisal tool (e.g. Jadad scoring) to look for bias in the study types. This suggests that they have systematically approached appraisal which is great.
having gone through this process there were only 8 papers to include. Considering the magnitude of the subject this is surprising. Only one RCT was found 6.
What about outcomes?
This is a key question and also influenced the choice of papers. The studies had to report patient focused outcomes such as mortality and neurological outcome. This is good and much better than looking for lesions at angiography for example. We want to know about patient focused outcomes and not interim findings. This is great, but did limit the number of included papers. Not all papers reported early and late mortality and not all reported neuro outcomes. This means that the meta-analysis around each outcomes included 4-6 papers only.
What are the principle results?
Overall there appers to be a benefit for early angiography. In terms of hospital mortality there was a statistically significant difference in outcome in favour of early intervention, death was 19.6% vs 35.6% (OR = 0.46, 95% CI = 0.36–0.56, P < 0.001).
For good neuro outcomes there was also a benefit for early intervention 34.8% with early intervention vs. 27.5%. Early angiography was also shown to be associated with improved neurological outcomes on discharge (OR = 2.00, 95% CI = 1.50–2.49, P < 0.001) as well as on follow-up (OR = 1.48, 95% CI = 1.06–1.90, P < 0.001).
These are pretty large effect sizes. The absolute risk reduction for mortality is 16% and so the number needed to treat (NNT) is 6.5. That’s amazing, but read on for cautions on that figure…
So we should send everyone to cath lab then?
Well hang on a second. There are some significant cautions here. Many of the studies included in this meta-analysis are retrospective and thus prone to significant bias. In my experience the decision to take non ST elevation patients to the cath lab is a judgement call with our cardiology colleagues. In other words there is a significant selection bias in who they take to cath lab immediately. It depends on things like the time of day, the availability of services and who is on. It is entirely plausible that decisions to take patients early to the cath lab are based on a perception of a likely need for intervention with a positive outcome. RCTs are a way of avoiding this, but the only RCT in this analysis was small and had a non-significant result. That may be telling as retrospective cohort studies have a habit of over estimating treatment effects.
We should also be mindful that there are several reasons why patient may arrest. For example a patient with Brugada7, ARVD8 or long QT syndrome9 would not obviously benefit from immediate coronary angiography. Such findings might be apparent on post ROSC ECGs and thus an informed decision might be made to not proceed to angiography. We cannot then just assume that a cardiac arrest is a result of ischaemic heart disease. As I often say to my patients, with respect to the heart we can have problems with the plumbing (IHD) or the electrics (arrythmias), and you wouldn’t want a plumber to sort out your electrics. Now I know that’s a little simplistic and my cardiology colleagues are expert in both, but hopefully you get my point. There is a pathophysiological reasons to not recommend immediate angiography in all patients.
So what next?
I think this paper supports my belief that patients whom I believe have IHD should go to the cath lab, but I’ll admit that the strength of evidence is not that great. I think we do need to make this an informed decision and not a knee jerk response (so we need to have sensible conversations with our interventional cardiologists). If in doubt, I’d still refer for early angiography and at the very least would transfer the patient to a cardiology centre. The NNT is very impressive, but may well be a result of significant bias in the trial designs. It might be that high, but somehow I doubt it.
Of course, what we really need for a definitive answer is an RCT conducted amongst an appropriate group of patients. Interestingly the only RCT in this analysis is described as a pilot trial and so we may just get it. Until then I will be picking up the phone and referring the majority of patients.
vb
S
@EMManchester
Before you go please don’t forget to…
- Subscribe to the blog (look top right for the link)
- Subscribe to our PODCAST on iTunes
- Follow us on twitter @stemlyns
- See our best pics and photos on Instagram
- PLEASE Like us on Facebook
- Find out more about the St.Emlyn’s team
References


Hello Simon,
Great find and read…What was the definition of early PCI? <6hrs, <12hrs, <24hrs? I find that in STEMI, we shoot for that magical 90min mark but in NSTEMI we often see delays out to 24hours. 100% agree with consulting your cardiologist in these cases.
Couple of points:
1. ACS is a dynamic process. Be sure to look at the pre-hospital ECG, as you may see STEMI compared to the ECGs you get in your department
2. 25% of NSTEMIs are known to be total (100%) occlusion of coronary arteries, so if a patient is having continued chest pain, hemodynamically unstable, or extremely "elevated" troponin level (i.e. not the troponin of 0.1 or 0.2), I would actually push for a sooner than later cath (i.e. <2hrs)
My 2 Cents for what its worth
Salim
Thanks Salim, in terms of early/emergent vs late then this was defined differently across trials (another bias of course that often crops up in retrospective analysis).
So in the paper they state that early CAG intervention ranged from on admission, within 2 h of admission, or between 6 and 12 h after hospital admission.
That’s a fairly broad window, and one might argue that the later times would not be as emergeny as we consider in Virchester (which would more be the within 2h window). If anything I suspect the effect size would be greater if only very early trials included – but of course I have no evidence to support that and it’s entirely belief and not evidence.
S
Prof,
Am teaching on this very topic soon on a p-h MSc. Thank you for your excellent summary I’ll be referring to it.
Literally just writing our post cardiac arrest pathway based on resus councils – states ST elevation for angio – no mention of LBBB , Wellens etc ( which,incidentally I got a patient with Wellens/ LAD occlusion across to the cardiologists last week with moderate pain ) so I’m going to now have to rethink this bit of the pathway! Maybe discuss all with PCI centre if suspected IHD? What do you think?
Pingback: Papers of December ’17 – The Resus Room
Pingback: JC: Do early pupil responses predict outcome in cardiac arrest? St Emlyn's - St.Emlyn's
Pingback: JC: Do early pupil responses predict outcome in cardiac arrest? St Emlyn's • St Emlyn's